When it comes to vacuum aluminum brazing there are a lot of advantages over other metal-joining processes (strength, tighter control, purity of connection joints, etc.), but you also need to know the details on how to properly braze your parts.

1. Make sure your parts are clean
First, you need to make sure your parts clean and free of stamping and cutting oils. Typically, this is achieved by either chemical or mechanical cleaning. Although, in the case of mechanical cleaning, be sure that the proper surface roughness is maintained, which helps to ensure a solid braze. After that, it’s all about time and temperature.
2. Know your process cycle
A typical vacuum aluminum brazing cycle is relatively short due to the fast pumping and heating characteristics of the vacuum furnace, as well as the high thermal conductivity of the aluminum parts being brazed.
Typical brazing process cycle:
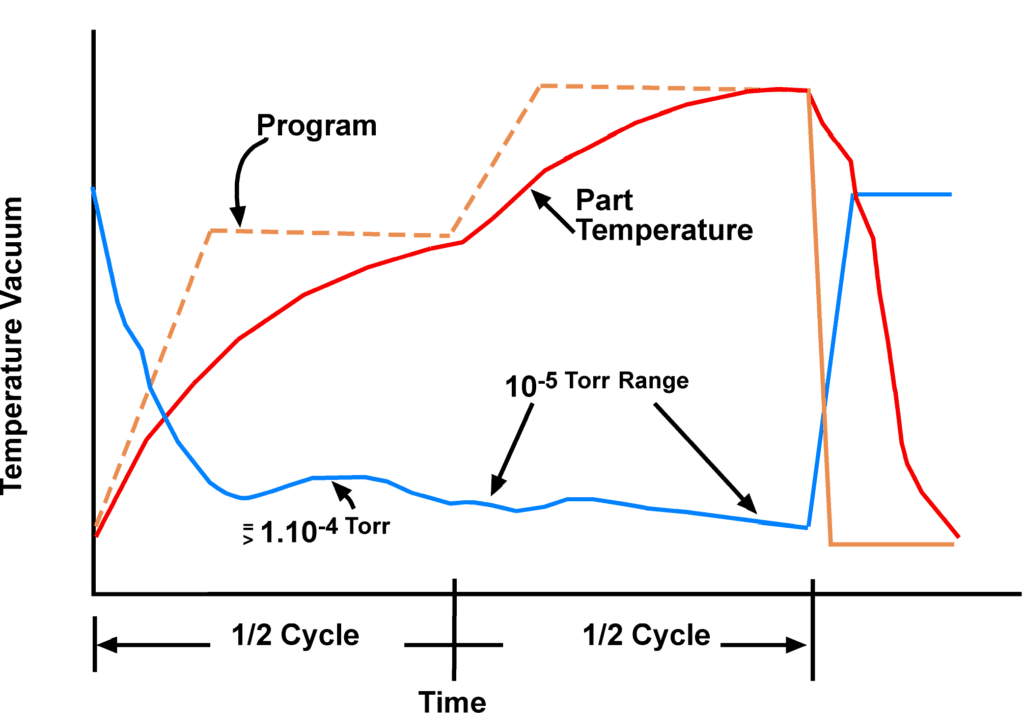
3. Ensure fast pumpdown time
The vacuum pumping capacity must be adequately sized to minimize the pumpdown time of a new load to a deep vacuum level. This helps initiate the heating cycle and create a proper throughput, which ensures you keep up with the significant outgassing taking place during the heating cycle due to magnesium vaporization. The deep vacuum level is a critical process parameter because it ensures a pure environment for brazing.
Changes in purity levels in relation to various vacuum levels:
| Pressure in mbar | Total Vol.-% | O2 Vol.-% | N2 Vol.-% | O2 ppm | N2 ppm |
| 1013 | 100 | 20 | 79 | 200*103 | 790*103 |
| 1 | 0.1 | 0.026 | 0.1 | 264 | 1040 |
| 10-1 | 0.01 | 0.0026 | 0.01 | 26.4 | 104 |
| 10-2 | 0.001 | 0.00026 | 0.001 | 2.64 | 10.4 |
| 10-3 | 0.0001 | 0.000026 | 0.0001 | 0.264 | 1.04 |
| 10-4 | 0.00001 | 0.0000026 | 0.00001 | 0.026 | 0.1 |
4. Use magnesium
It is necessary to use magnesium as an additive to the filler metal and/or the base metal of the parts for the following reasons:
- Magnesium starts to vaporize at around 1,058 °F (570 °C), and then acts as a “getter” for oxygen and water vapor, thus improving the purity of the brazing vacuum.
- Magnesium reduces the alumina oxide that exists on the surface of aluminum to promote uniformly accelerated wetting of the joint surfaces.
The following reactions occur during the vacuum brazing process:
Mg + H2O → MgO +H2
Mg + O2 → 2 MgO
3Mg + Al2O3 → 3MgO + 2Al
Mg + N2 → Mg3N2
The vaporization of the magnesium in a vacuum environment, also known as a “mag burst,” produces heavy outgassing for a short period of time. However, the slower the heating rate, the smaller the magnesium vaporization rate. Due to this gas load, the vacuum pumps must be adequately sized to maintain a good working vacuum (10-4 to 10-5 Torr range).
5. Maintain temperature uniformity
Second to deep vacuum level, precise temperature control and uniformity are also required to achieve a successful braze. An acceptable temperature uniformity during a brazing cycle is ±5 °F (3 °C) of set point. Aluminum brazing has a very narrow window of acceptable brazing temperatures because the filler metal has to liquidize before the base metal reaches its solidus temperature. This temperature difference can be as small as ±10-18 °F (6-10 °C).
Therefore, it is necessary to use a heating step at a soak temperature just below the solidus point of the filler metal to ensure that all the parts and joints to be brazed reach the correct temperature at about the same time. At this time, the ramp to brazing temperature should start so that the filler metal begins to melt and capillary wetting of the braze joints occurs.
The time duration at brazing temperature must be kept to a minimum as the melted filler metal is vaporizing in the deep vacuum and trying to wet the braze joints. Too much loss of filler metal to vaporization results in poor joint wetting and subsequent loss of joint strength and sealing ability.
After the brazing temperature soak duration is complete, it is followed by an immediate vacuum cooling cycle, which solidifies the filler metal in the braze joints and stops the vaporization of material.